It seems that it’s rare for a week to go by that we don’t hear about – or even experience – yet another pet illness or reaction to animal food, drugs, vaccines, or pesticides. At times, Whole Dog Journal’s articles and blog posts will include the advice to “report any adverse events.” And it’s excellent advice – so here’s when, how, and why you should report these events.
Adverse Events Defined
Animal drugs, vaccines, and pesticides are subjected to tests to establish their safety and efficacy. However, this evaluation process is typically conducted on a relatively small number of animals prior to being approved and marketed. As a result, there is potential for previously unobserved problems to emerge after the product is approved, goes on the market, and is administered to a larger population of animals.
An adverse event (AE) in animals is any unfavorable or unintended occurrence that happens during or after use of an animal drug or veterinary product or device. Suspected lack of efficacy and reactions by humans exposed to the product or treated animals also qualify as adverse events.
Adverse Events or Side Effects?
It is important to clarify the difference between adverse events and side effects. Side effects are secondary undesired effects that may occur when using a specific drug and have been shown to be associated with the product by scientific studies. Side effects are tracked and investigated extensively during clinical trials before entering the market. In contrast, adverse events are not consistent with applicable product information or characteristics of the drug.
It is important to consult with your veterinarian about potential side effects associated with a product prior to use. For example, a lump forming at the site of a vaccination is a known side effect; it is not an adverse event. If your dog does experience a serious issue after a vaccine, the administration of a drug or pesticide treatment, or eating pet food or treats, contact your veterinarian or veterinary emergency clinic immediately.
Reporting side effects is still a good idea, however. Even though side effects may be known, reporting these can be an effective means of heightening awareness of a particular issue with the product and could possibly lead to additional product review, especially if the side effects are serious.
When Should You File a Report?
One of the responsibilities of the U.S. federal government is ensuring the safe and healthful use of products within the United States. If your pet experiences a serious side effect or an adverse event, a report should be filed with the appropriate governing agency.
If you’re uncertain about whether you should file a report or the process, ask your veterinarian for guidance. Often veterinarians themselves will handle it; if this occurs, there is no need for you to report the incident. If your veterinarian has not reported the AE or was not involved, you should make the report yourself. In either case, it is important to report it as soon as possible, as timely reporting enables agencies to take any necessary action quickly.
There are several government agencies that oversee the reporting of veterinary adverse events and the process varies with each department. Regardless of the government agency involved, the manufacturer of the product should also be notified of the adverse event.
Animal Drugs
All veterinary drugs (and devices) are regulated by the Food and Drug Administration’s (FDA) Center for Veterinary Medicine (CVM) under the Food, Drug, and Cosmetic Act. New veterinary drugs must first be approved for use by the CVM. The CVM evaluates the product and establishes the safety, effectiveness, and conditions of use; this information is required to be on the product labeling. After approval, the CVM will continue to monitor the use of these products to ensure that they remain safe and effective; this monitoring activity is referred to pharmacovigilance.
Thousands of drugs are administered to pets every day. There are often side effects, but hopefully those are known, minor, and temporary. When a pet has an unexpected reaction to a veterinary drug, it is important to file an Adverse Drug Event (ADE) report. This report can be filed directly with the FDA by pet owners.
There you will find the link to download the PDF Form FDA 1932a, “Adverse Experience, Lack of Effectiveness or Product Defect Report.” This form is used to report adverse drug experiences for any animal drug (whether the drug is FDA-approved or not).
The CVM requests that the report be as detailed as possible and include everything from the brand of food and treats your pet eats, to any supplements your pet is given, along with your pet’s medical history (including names and amounts of all drugs, information about any recent surgeries or procedures, veterinary test results and examination findings), as well as any other relevant information. The more details you provide, the more information the CVM has available to research the issue. Upon completion, email the form to CVM1932a@fda.hhs.gov.
Both the CVM and the American Veterinary Medical Association (AVMA) recommend also reporting any adverse drug events directly to the manufacturer. Manufacturers are required to report any adverse drug experiences to the FDA. The drug manufacturer’s information is usually on the product packaging, including prescription bottles received from the pharmacy. You may also report the event directly to the CVM.
Pet Food and Treats
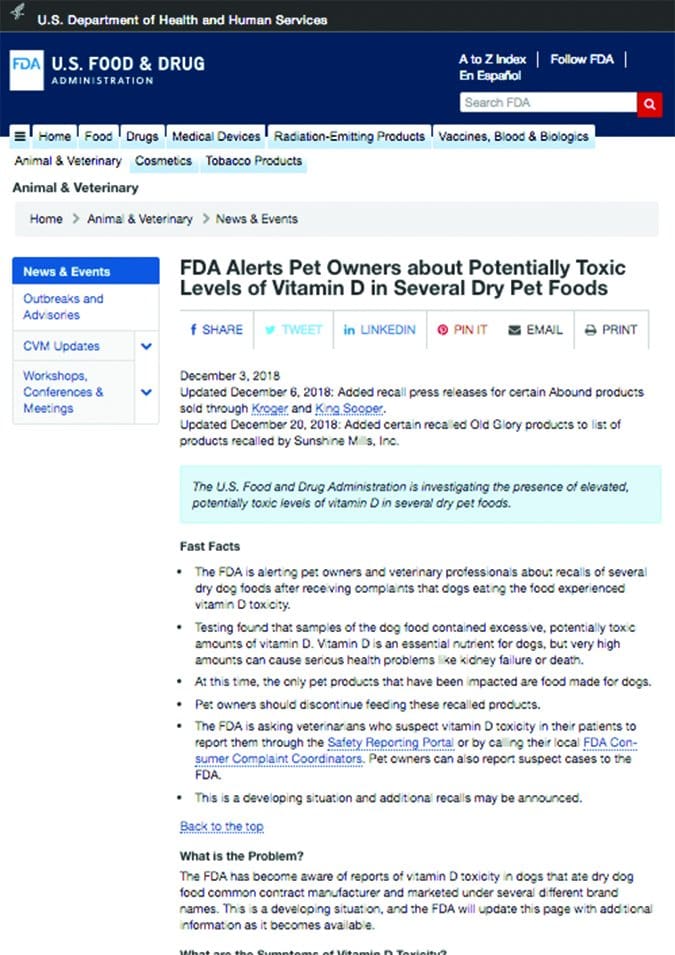
Adverse events related to pet food and treats also fall under the CVM’s jurisdiction. The responsibilities of the CVM include ensuring that pet food “is safe, made under sanitary conditions, and properly labeled” as well verifying that a “food additive for use in food for animals is safe and effective before approving it.” Reports regarding pet food and treats can be filed electronically using the CVM’s Safety Reporting Portal.
Problems such as mold or foreign objects in food should also be reported as they can pose a health and safety issue for other pets.
The other option for reporting issues with pet food and treats is to call your state’s FDA Consumer Complaint Coordinator. You can use this link to find your representative.
Before you call or start your online report, assemble all the relevant data. A detailed list of the information you should have on hand to complete your report is listed here.
Animal Vaccines
Vaccines for pets are regulated by the U.S. Department of Agriculture via the Center for Veterinary Biologics (CVB). The CVB is involved with the regulation of products that diagnose, prevent, or treat animal diseases as well as ensuring that veterinary biologics, such as vaccines, are safe and effective. Detailed information about reporting vaccine-related AEs can be found here.
The preferred method for reporting an adverse event associated with a vaccine is through the CVB’s online reporting system.
Reports may also be submitted by telephone by calling the CVB at (800) 752-6255, or by downloading the Adverse Report Form and faxing it to (515) 337-6120, or sending via mail to Center for Veterinary Biologics, 1920 Dayton Avenue, PO Box 844, Ames, Iowa 50010.
Again, it is recommended that vaccine-related adverse events also be submitted directly to the vaccine manufacturer. In addition to simply taking the report, many of these manufacturers have in-house veterinary services departments that not only receive such reports but also may be able to provide diagnostic advice, treatment recommendations, and guidance on product use.
Pesticides
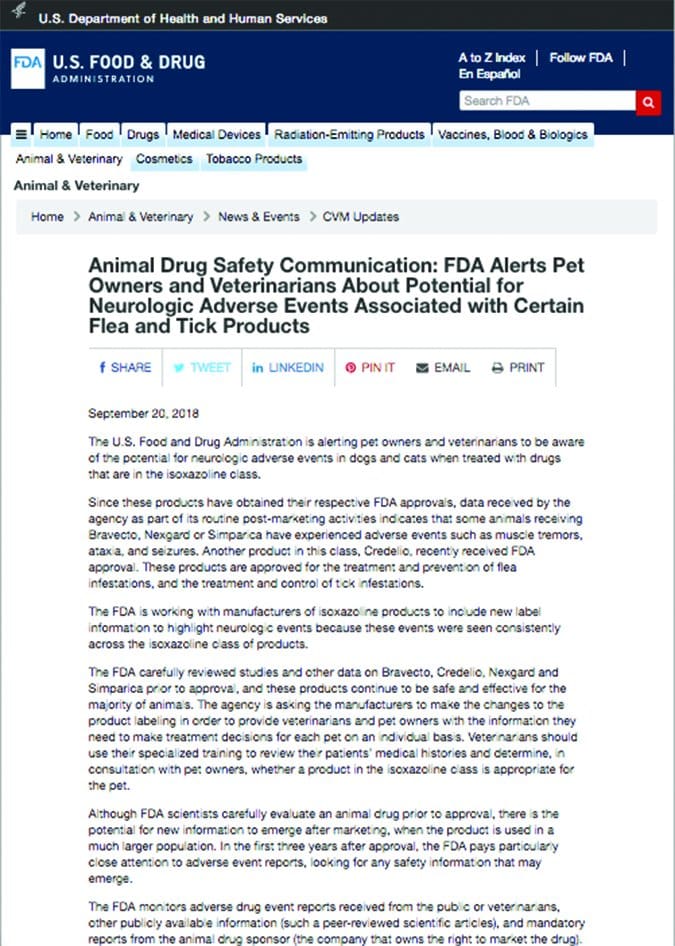
There are a number of pest-control products used in association with our pets, especially for eradication of ectoparasites and insects. The FDA regulates the flea and tick products that are given orally (making them qualify as a medication), including Bravecto, Nexgard, Simparica, and Credelio.
If a product is regulated by the FDA, it may be labeled with the statement “Approved by the FDA” followed by a six-digit New Animal Drug Application (NADA) number or, for generic drugs, an Abbreviated New Animal Drug Application (ANADA) number on the packaging or label. If the product’s registration number does not appear on the label, it may take some research to find the number; it’s not required to be present on the label.
Adverse events associated with oral medications that kill internal or external parasites may be reported by the same process as animal drugs (see above), using form FDA 1932a, “Adverse Experience, Lack of Effectiveness or Product Defect Report.”
Topical products, however, are regulated by the Environmental Protection Agency (EPA), under the Federal Insecticide Fungicide and Rodenticide Act. These can be identified by the EPA Registration Number (sometimes written as EPA Reg. No.) printed on the label.
Look for an EPA registration number on the package, usually near the manufacturer’s information. Adverse events associated with these products, as well as other pesticides, are reported to the National Pesticide Information Center (NPIC).
However, the NPIC electronic reporting system is restricted for use by qualified professionals, such as veterinarians or their staff. Pet owners can still report an adverse effect by a pesticide on their pet by calling the NPIC directly at (800) 858-7378 and filing a report over the phone.
What Happens with the Reports?
The governing agencies all have differing approaches to handling Adverse Event Reports, but in general, these reports are reviewed and evaluated for risks to animal (and/or public) health.
Every report is important; if the AE is assessed to be of concern, the initial review leads to follow-up activity. Products that have caused or may cause a serious illness, injury, or a life-threatening situation are given priority. While monitoring continues indefinitely, the FDA pays particularly close attention to adverse event reports submitted in the first three years following approval of a product.
The CVM has created a database – the Cumulative Adverse Drug Event Summaries Report – “so that veterinarians and animal owners can have easily available access to information about signs that have been associated with drugs.”
The database lists the numbers of ADEs received for a particular drug, by species, and route of administration. Sounds great! But at present, the database contains only the cumulative summaries of the data received on paper reports for the period between 1987 and April 30, 2013. ADEs have been submitted electronically since then, but the task of collating the paper reports with the electronically submitted reports is not yet complete.
The plan is to eventually have the database brought current and updated monthly – but we aren’t holding our breath. A note on the webpage above says, “We anticipate having updated adverse event information available in late 2017 or early 2018.”
Why Report Adverse Events?
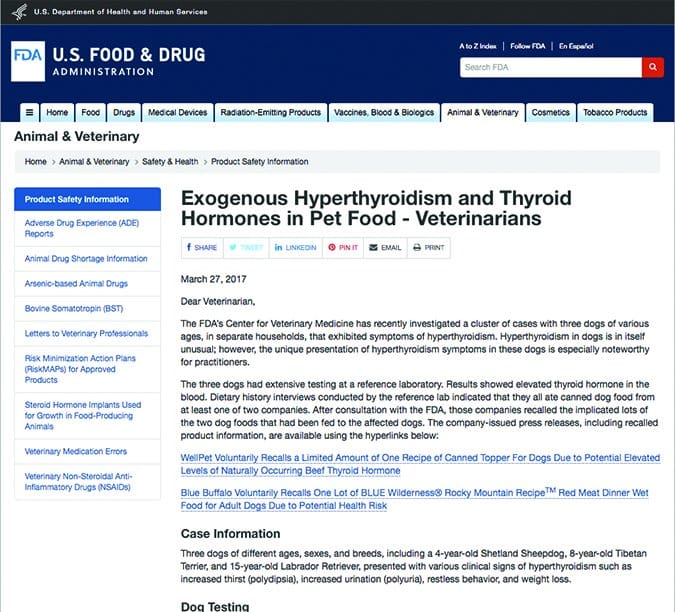
In March 2017, the CVM distributed a “Dear Doctor” letter to veterinarian professionals in response to their findings from the examination of three reported cases of hyperthyroidism in dogs of varying ages and in separate households that were fed canned dog food from at least one of two companies (FDA Dear Doctor letter, “Exogenous Hyperthyroidism and Thyroid in Pet Food,” March 27, 2017).
The CVM’s research into these three cases found that it warranted widespread notification and the resulting letter detailed their research, findings, and actions. It was determined that the pet food contained exogenous sources of thyroid hormone, capable of causing clinical signs. The manufacturer recalled the implicated lots of the two foods that had been fed to the affected dogs. (The health of the dogs improved after eating different food.)
By reporting AEs and other problems experienced with animal products, pet owners, veterinary professionals, and manufacturers play a valuable role in maintaining health and safety. Reports act as an alert system, not only for the governing agency, but also for pet owners and veterinary professionals. The government, manufacturers, and veterinarians receive critical information to determine if further investigation is warranted.
When it happens to our pets, naturally we feel it is warranted. As evident in the example above, the reporting of just three cases resulted in an investigation and recall, demonstrating that reporting is effective and can contribute to our pets’ health and welfare.
Investigations triggered by AEs can lead to changes in a product’s labeling or use, the release of safety warnings and communications, product recalls, and even the rescinding of agency approval or permanent withdrawal of a product from the market. Again, reporting adverse events can be invaluable.
The reporting systems do have some deficiencies. Being “passive surveillance systems,” there is no active search for cases but rather the reliance on obtaining initial data from the reports submitted. This type of system can suffer from under-reporting (reports aren’t submitted) or over-reporting (reports submitted are not related to the suspected product). Furthermore, receipt of a report does not necessarily mean that the product caused an adverse event, or even that a particular event actually occurred.
If your pet experiences an adverse event, seek veterinary treatment. If you suspect it is due to a drug, food or treats, vaccine, or pesticide, you may want to consider obtaining special diagnostics, such as having a pet food tested by a private laboratory. Be aware, however, that these expenses will not be covered by the regulating agency, but may assist in the treatment of your pet and add valuable data to your report.
Barbara Dobbins, a former dog trainer, writes about dogs and studies canine ethology. She lives in the San Francisco Bay area with her dogs, Tico and Parker.