[Updated July 18, 2018]
CUSHING’S DISEASE IN DOGS: OVERVIEW
1. Don’t dismiss changes in your dog as normal aging. Your veterinarian may find an underlying, treatable cause.
2. Treatment for Cushing’s can improve quality of life for your dog. Don’t be afraid to try drugs when needed.
3. Watch closely for signs such as lack of appetite that may indicate drugs need to be stopped or dosage adjusted.
4. lf your dog has all the signs of Cushing’s but tests are negative, consider atypical Cushing’s or adrenal exhaustion as a possible cause.
Sometimes life-threatening illnesses develop so slowly that no one pays much attention. An older dog who’s always thirsty drinks huge quantities of water. He’s hungrier than usual and has developed a thin coat and pot belly. He pants all the time, and now his hind legs are losing muscle tone and getting weak. Many owners dismiss these as normal signs of aging, but they are classic symptoms of Cushing’s disease, which affects an estimated 100,000 dogs in the U.S. every year.

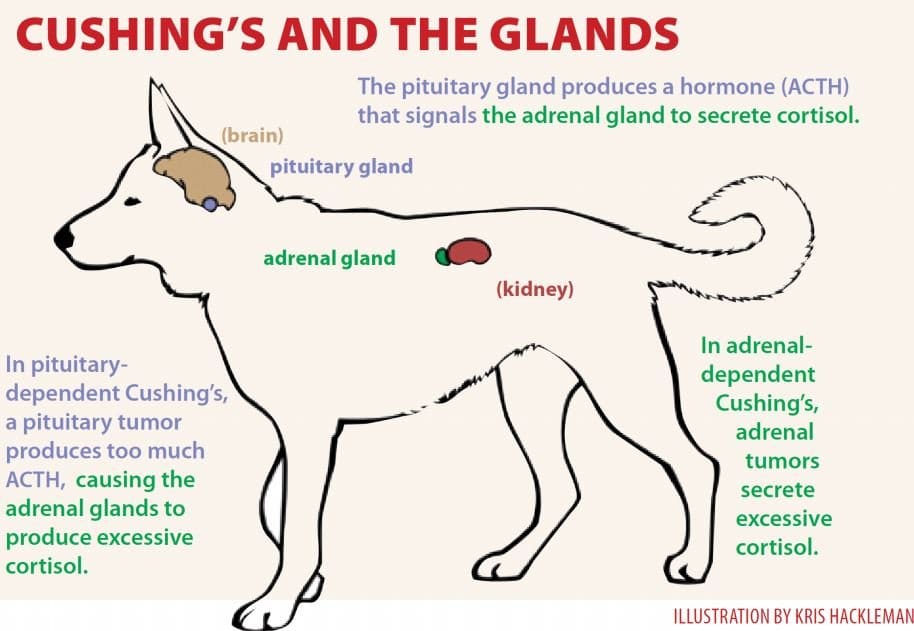
Also known as hyperadrenocorticism, Cushing’s is the opposite of Addison’s disease. Both illnesses involve the adrenal glands, which produce cortisol – a stress hormone that helps prepare a dog for fight or flight by breaking down muscle and fat to convert into glucose. While Addison’s disease results from insufficient cortisol, Cushing’s disease results from too much – and prolonged exposure to cortisol can have negative effects on the entire body. Cushing’s puts dogs at increased risk for diabetes, pancreatitis, calcium oxalate bladder stones, pulmonary thromboembolism, hypertension (high blood pressure), urinary tract infections, protein-losing kidney disease, hair loss, skin problems, and congestive heart failure.
Cushing’s disease affects primarily middle-aged and older dogs. The majority (about 85 percent) of Cushing’s disease cases are caused by tumors of the pituitary gland; these are usually benign (adenomas). About 15 percent of Cushing’s cases are caused by tumors of the adrenal gland; about half of these adrenal tumors are benign and about half are malignant (carcinomas).
In pituitary-dependent hyperadrenocorticism (PDH), tumors cause the pituitary gland to overproduce adrenocorticotropic hormone (ACTH), which signals the adrenal glands to release cortisol. The average age at diagnosis with PDH is 10 years, and the most commonly affected breeds are Poodles, Dachshunds, Beagles, German Shepherds, Boxers, Boston Terriers, Labrador Retrievers, and terriers, with 75 percent of patients weighing less than 44 pounds.
With adrenal-dependent Cushing’s, the adrenal tumor itself secretes excessive cortisol. The average age for diagnosis with adrenal tumors is 11 years, with Poodles, German Shepherds, Dachshunds, Labrador Retrievers, and terriers predominating. About half of all cases occur in dogs weighing more than 44 pounds, and females may be more commonly affected.
Kris Hackleman
Pituitary Cushing’s Disease in Dogs
Girl, a Miniature Schnauzer, has lived with Cindy Bright of Indiana, Pennsylvania, since her first owner passed away in 2006. Girl is famous for eating anything (including dehydrated or living toads); at 27 pounds, she was seriously overweight.
By February 2008, Girl’s symptoms included excessive thirst and urination, overeating, urinary incontinence, and occasional hyperexcitement or aggression. She was diagnosed with PDH and was given Vetoryl (trilostane), a drug that was not approved in the U.S. until 2009 for the treatment of both pituitary- and adrenal-dependent Cushing’s, but which has been used in the U.K. since 2001. Bright imported the drug from the U.K. (something that could be accomplished with a prescription and by filling out a form to obtain permission from the FDA).
Today Girl is doing well, and her weight is a normal 19 pounds. Her occasional health setbacks tend to follow dietary indiscretions, such as her consumption of a 6-pack of English muffins that someone set outside the basement freezer and her indulgence in wild rabbit droppings, which Bright describes as Girl’s “caviar.” Girl suffered a serious bout of pancreatitis in 2010. “Life is like walking the tightrope,” Bright says. “One slip and it’s difficult to get everything back on course.”
Like Girl, all dogs with Cushing’s disease require careful monitoring. Depending on the drug used to control symptoms, even minor changes in condition or behavior can signal major problems, and conscientious caregivers keep a close eye on anything that seems unusual or different.
In Springfield, Vermont, Kathy Mason’s Beagle, Chance, was diagnosed with PDH at 8 years old. He was treated with Anipryl, which was not effective, and then switched to Lysodren (mitotane). “After about two years of good bloodwork,” says Mason, “Chance went into an Addisonian crisis. We thought we were going to lose him, but he survived and stayed Addisonian for the rest of his life. It was so much easier to treat. He passed away in January, five years post diagnosis, at the age of 14 years old.”
While most pituitary tumors are microscopic, 10 to 20 percent are larger than 10 millimeters (3/8 inch) in diameter. Called macrotumors or macroadenomas, almost all of these pituitary tumors are benign, but about half are large enough to compress the brain and cause neurological symptoms such as listlessness, poor appetite, confusion, walking in circles, or seizures. Most dogs are diagnosed with Cushing’s at least six months before neurological symptoms develop. Breeds with short noses, such as Boxers and Pugs, tend to have the largest pituitary tumors.
Most dogs with pituitary tumors are treated with medication because their tumors are considered inoperable. However, a rather new surgical technique allows some tumors to be removed.
Another option for large pituitary tumors is radiation therapy, which is expensive ($3,000 to $5,000 in the Los Angeles area) and can have adverse side effects such as skin and hair loss, hearing loss, or problems with tear production in the eyes. Although successful in 70 percent of cases, further treatment for Cushing’s is usually required, and tumors treated with radiation are likely to recur. Survival time is not increased if neurological problems are severe.
Adrenal Cushing’s Disease
hree years ago Maggie, a 12-year-old black Labrador Retriever/hound-mix in Atlanta, Georgia, developed a voracious appetite, drank incredibly large amounts of water, and began raiding the trash. Her owner, Kristin Boggs, took Maggie to their veterinarian, who suspected Cushing’s. “Usually Cushing’s is pituitary-based,” she says, “but instead of assuming this, my vet ordered an ultrasound test. He had once before seen a case of Cushing’s that was caused by an adrenal tumor. Sure enough, Maggie had a large tumor on her adrenal gland.”
The solution was to remove the entire gland. Maggie’s surgery took place at a special internal medicine veterinary clinic, where a post-surgical nurse attended her through the night.
“Maggie came through with flying colors, despite an incision almost the full length of her belly,” says Boggs. “She’s a tough old girl. Thankfully the tumor was benign. After a few weeks of medication to balance her cortisol levels, she was back to normal.”
In Toledo, Ohio, Linda Boyle’s Springer Spaniel, Cody, was diagnosed with Cushing’s disease at age 11 after developing a ravenous appetite and gaining weight. Cody’s ultrasound exam revealed tumors on both adrenal glands.
“A bilateral adrenalectomy is very risky,” says Boyle, “but it worked well for Cody. He became an Addison’s dog because his body no longer produced any cortisol at all, but he lived another two years with good quality of life before passing away from what we believe was pneumonia.”
Because Cushing’s patients tend to have high blood pressure and poor healing ability, several months of medical therapy may be needed to strengthen the patient prior to surgery. After surgery, prednisone or other corticosteroids may be required for a few months until the dog’s ability to produce cortisol recovers, and some dogs may need mineralocorticoid replacement as well.
In a survey of 63 dogs who had surgery, 4 (6 percent) had inoperable tumors and were euthanized; 18 (29 percent) died during surgery or soon after because of complications; and the average post-surgical life span for the remaining 41 dogs was 46 months.

As this article goes to press, Maggie’s Cushing’s symptoms have returned, caused by a new tumor on her remaining adrenal gland. “I can’t believe this has happened to us again,” says Boggs. “It’s an exact repeat of what we went through three years ago. We don’t know if the tumor is malignant or benign, as we don’t feel extensive surgery at her age (15 ½) is reasonable. My vet and I have decided to treat her with trilostane and see how she does. My priority now is that she is happy and comfortable.”
Diagnosing Cushing’s Disease in Dogs
Cushing’s disease isn’t always easy to diagnose. Routine test results that can point toward Cushing’s include:
- High or very high alkaline phosphatase (ALP, ALKP, SAP). Although this is a “liver enzyme,” marked elevation is associated with cortisol. It does not indicate liver disease if other liver enzymes such as ALT (alanine aminotransferase) are normal.
- Increased neutrophils and decreased lymphocytes (stress leukogram).
- Elevated cholesterol and triglycerides due to abnormal fat mobilization.
- Fasting hyperglycemia due to insulin resistance.
- Liver enzymes may be mildly elevated.
- Dilute urine (low specific gravity) due to excessive drinking.
- Protein in urine (microalbuminuria or proteinuria).
- X-rays may show an enlarged liver and occasionally calcium deposits in the skin (calcinosis cutis, which feel like rocks and cause itching).
Dogs with Cushing’s are prone to bladder, skin, and other infections due to the immunosuppressive effects of cortisol. About 20 percent of dogs with Cushing’s have an inapparent bladder infection, requiring a culture (not just urinalysis) to detect.
Confirming a Cushing’s diagnosis can be challenging, especially as false positives are common in dogs with nonadrenal illness. The following tests are commonly used:
- Urine Cortisol:Creatinine Ratio (UCCR) is a screening test only. A negative result rules out Cushing’s, but anything that increases stress can cause a positive result, necessitating further testing. Use urine samples caught at home for this test so that the stress of a vet visit doesn’t influence the results.
- Low Dose Dexamethasone Suppression (LDDS), considered the most accurate for diagnosing Cushing’s, takes eight hours in the hospital. A baseline blood cortisol level is measured, then a tiny dose of dexamethasone (corticosteroid) is injected. In a normal dog, blood cortisol levels drop after eight hours. Blood samples are sometimes checked after four hours, as this can help classify the form of Cushing’s. About 95 percent of dogs with Cushing’s test positive, but false positives are also common, and the more severe a nonadrenal illness, the more likely a false positive result. This test is preferred if an adrenal tumor is suspected.
- ACTH Stimulation: No longer recommended for diagnosis, as its sensitivity is poor, its cost is high, and it cannot differentiate between pituitary and adrenal forms. This test is more specific than LDDS, however, meaning that strongly positive results definitely indicate Cushing’s disease, and false positives are unusual, but false negatives are common.
This test is useful for monitoring treatment, and it is the only test that can be used for dogs currently being treated with corticosteroids. A low-normal or blunted response indicates iatrogenic Cushing’s (see sidebar, facing page). This may be the preferred test if symptoms are mild, for it is better to miss the diagnosis than get a false positive.
Test results must be considered in combination with symptoms. A dog with at least three of the typical signs plus a positive test is considered to have Cushing’s, but a dog with no signs but maybe high ALP and a positive test may not, in which case it’s best to take a wait-and-see approach. If signs are present but test results are negative, consider retesting in three to six months or test for atypical Cushing’s.
Different Types of Cushing’s Disease
Classifying the type of Cushing’s disease as pituitary or adrenal helps determine which treatment is best. The endogenous ACTH level is felt by many to be the most accurate method of identifying the type of Cushing’s, but this test is difficult to run, as serum must remain frozen during transport to the lab. Dogs with pituitary tumors have high ACTH levels, while those with adrenal tumors will have low or no measurable ACTH.
LDDS may be used for classification when a 4-hour sample is drawn. If the 8-hour sample is > 30 nmol/L and the 4-hour sample is < 30, or both are less than half of the baseline sample, the pituitary form is likely.
The high dose dexamethasone suppression (HDDS) test is similar to LDDS, but a high dose of dexamethasone is used. This test is done only on dogs already known to have Cushing’s. Dogs with a pituitary tumor will show suppression in circulating cortisol levels 75 percent of the time, but those with adrenal tumors never show suppression.
Adrenal tumors can be seen on ultrasound 30 to 50 percent of the time. An adrenal tumor is suspected whenever one adrenal gland is observed to be enlarged while the other is not visible. If they have not spread, adrenal tumors should be removed, if possible, but surgery is difficult and usually requires a specialist.
Two normal or large adrenal glands indicate the pituitary form of Cushing’s, although enlarged adrenal glands are also often seen in cases of nonadrenal disease due to the chronic stress of the illness. An MRI or CT scan can detect pituitary tumors and determine their size. Two-thirds of dogs with tumors greater than 10 millimeters initially will develop neurological signs and may benefit from immediate radiation therapy, even before signs are seen.
Medical Treatment for Cushing’s Disease in Dogs
The best treatment for Cushing’s disease depends on the severity of symptoms, type of Cushing’s, risk of adverse effects, and cost factors. Medical therapy is used to control clinical signs, but it does not cure the disease. It is not likely to prolong life but does improve the dog’s quality of life. Not treating is also a viable option, particularly if symptoms are minimal. In this case, complementary care may be especially useful.
If the dog suffers from a concurrent medical condition, it is important to treat and stabilize that illness if possible before treating for Cushing’s. Treatment for dogs with concurrent nonadrenal illness, such as diabetes, is complex and beyond the scope of this article.
Mitotane
Mitotane (Lysodren®), which is used for both pituitary and adrenal Cushing’s, was the only treatment available until recent years. Mitotane is convenient and relatively inexpensive, but it has potentially severe side effects. A chemotherapy drug, it works by selectively eroding the two inner layers of the adrenal cortex that produce cortisol while tending to preserve the outer layer that makes aldosterone. If too much is eroded, the result is a cortisol deficiency.
About 30 percent of dogs treated with mitotane develop short-term reactions. If this happens, mitotane is discontinued and the dog is treated with prednisone until the adrenal glands recover. The destruction may be permanent, requiring treatment for atypical (or sometimes typical) Addison’s. Mitotane’s risk of a permanent or life-threatening Addisonian reaction is considered 2 to 5 percent.
Mitotane is given once or twice a day during the “induction” phase, during which adrenal erosion is induced. The drug must be given with food or it will not be absorbed. Mitotane should not be used for dogs with a poor appetite; inappetence indicates that something else is going on that must be diagnosed and addressed prior to beginning Cushing’s therapy.

During the induction phase, caregivers should notify their veterinarians as soon as their dog experiences any appetite reduction, decrease in water consumption, diarrhea, vomiting, lethargy, or listlessness. It is important to watch for these signs starting around the third day of treatment. These symptoms indicate that the end of the induction has been reached and it’s time for an ACTH stimulation test.
The induction time is variable, usually between 7 and 14 days, but some patients require just two to three days and others can take as long as six weeks, especially if they are being given other drugs that interact with Lydsodren, such as phenobarbital or insulin. If there is no response, dosage can be increased or mitotane can be reformulated in an oil base to increase absorption, or the drug trilostane can be used instead.
When the ACTH test and clinical signs indicate that sufficient adrenal erosion has taken place, daily use (induction phase) is stopped and lifelong “maintenance” treatment once or twice a week is begun. The ACTH test can be repeated after about a month, then one to four times a year thereafter.
If vomiting, diarrhea, appetite loss, or listlessness occur at any time during treatment, the dog may need a prednisone “antidote” pill, which should produce improvement within 30 minutes to two hours. Your veterinarian will probably prescribe prednisone and discontinue mitotane for a couple of weeks after that, before resuming at a lower dose. If there is no improvement, something else is causing the symptoms.
About half of the dogs treated with mitotane relapse at some point and require a second round of induction, and their maintenance dose may have to be increased.
Marked improvement should be seen within six to eight weeks of starting the maintenance dose, with a full reversal of clinical signs after four to six months of treatment. Usually the first improvements are more normal water consumption, urination, and appetite, and the last is hair regrowth. Muscle strength and exercise tolerance improve over the first three to four weeks.
Mitotane can be used to treat adrenal Cushing’s, but a higher dosage is needed, resulting in more adverse reactions. The average survival time in this case is 16 months.
Trilostane
Trilostane (Vetoryl®), used to treat both pituitary and adrenal Cushing’s disease, inhibits the enzyme 3-beta-hydroxysteroid dehydrogenase, which is involved in the production of several steroids, including cortisol.
Because trilostane can block aldosterone as well as cortisol, it can cause side effects such as dehydration, weakness, and electrolyte changes (low sodium and high potassium). In this case, the medication must be stopped temporarily; some dogs may also require fluid support.
For reasons that are not understood, trilostane occasionally causes acute adrenal gland necrosis, which can occur within days of starting the drug or after several months. The condition is unpredictable and is not dose-dependent. It requires prompt treatment with fluid therapy, glucocorticoids, and mineralocorticoids or it can be fatal (Addisonian crisis). This complication is permanent and irreversible, and lifelong supplementation of both mineralocorticoids and glucocorticoids will be necessary (i.e., treatment for typical Addison’s).
Trilostane is given once or twice a day with food. Side effects may be less common when given twice a day. Starting dosage is 1 to 3 mg per pound of body weight daily. Starting at the low end of that range is safest, even though the suggested starting dosage in the product brochure is higher.
Close monitoring in the beginning and after any dose adjustment is essential. Discontinue trilostane and contact your veterinarian immediately if signs of intolerance are seen. Common side effects include mild lethargy, vomiting, diarrhea, and appetite reduction, especially in the beginning. Most reactions are minor and are reversed by decreasing the dose or discontinuing therapy, but serious or fatal Addisonian reactions are also possible. Side effects should be followed by electrolyte and ACTH testing. Once symptoms resolve, the drug can be restarted at a 50 percent dose reduction.
Most dogs show decreases in drinking, urination, and appetite within one week, but some may take two months or longer. Trilostane controls clinical signs in dogs with adrenal tumors, but will not slow tumor growth.
Trilostane may be safer than mitotane, as side effects tend to be less severe and most are reversible. The risk of a permanent or life-threatening Addisonian reaction from trilostane is considered to be 2 to 3 percent.
Cost is trilostane’s main disadvantage, though monitoring costs may be less than with mitotane. Baseline cortisol levels measured 4 to 6 hours after trilostane administration may provide adequate monitoring without the need for ACTH stimulation tests.
Trilostane seems to predispose dogs to increased adrenal toxicity from mitotane, so it’s best to wait at least a month in between if switching to mitotane. When changing from another drug to trilostane, wait a couple of weeks or until clinical signs recur. This drug is not recommended for dogs with liver or kidney disease.
Ketoconazole
Ketoconazole (Nizoral®) is used to treat both adrenal and pituitary Cushing’s. This antifungal medication suppresses adrenal hormones in about 80 percent of dogs as a side effect.
A low dose is used for a week; then, if no side effects are seen, a higher maintenance dose is used. It is not possible to induce Addison’s disease with ketoconazole, so monitoring tests aren’t needed, other than after a month or so to determine if the medication is working.
Side effects can include vomiting and diarrhea, but no “antidote pill” is needed. Just discontinue until side effects go away, then restart at a lower dose.
Ketoconazole’s disadvantages are that it must be given twice a day indefinitely and its effect may be mild or temporary. This drug may reduce clinical signs in up to 30 percent of dogs with adrenal tumors, but side effects are common.
L-Deprenyl
L-Deprenyl (Anipryl® or selegiline) is used to treat pituitary Cushing’s only. It is also used to treat canine cognitive dysfunction.
L-Deprenyl works by inhibiting ACTH secretion via high dopamine levels in the intermediate pituitary area, where 30 percent of pituitary tumors are located, and may work only for dogs with such tumors. This drug has no serious side effects, but approximately 5 percent of patients experience minor nausea, restlessness, or reduced hearing. It cannot induce Addison’s disease, and no monitoring is required.
Because it’s difficult to determine whether the drug is working, and it may take considerable time to take effect, few veterinarians report good results. However, because some dogs improve and its safety margin is high, it may be worth trying in dogs with mild symptoms that are progressing slowly. L-Deprenyl can be combined with mitotane or trilostane for dogs with cognitive dysfunction, and it may increase the dog’s activity level and quality of life.
Anipryl is not recommended for dogs who also have diabetes, pancreatitis, heart failure, renal disease, or any other serious illness.
Freelance writer CJ Puotinen lives in Montana. She is the author of The Encyclopedia of Natural Pet Care and is a frequent contributor to WDJ.
Mary Straus is the owner of DogAware.com. She and her Norwich Terrier, Ella, live in the San Francisco Bay Area.







My little border terrier has atypical cushings disease. Her fur loss is heartbreaking. It’s taken me 2 and a half years to persuade my vet to test for Atypical as he wouldn’t do it. Finally he agreed and it’s positive- normal cushings test came back negative
What is the treatment for atypical and is it going well?
26,186 Satisfied Customers
✕
Virtual Assistant avatar
Pearl Wilson, Veterinarian’s Assistant
Welcome! What’s going on with your dog?
You
Hello
Virtual Assistant avatar
Pearl Wilson, Veterinarian’s Assistant
Hi. How can I help?
You
My dog is almost 10. He’s a rot put lab mix. Over the last 3 days he has been showing sudden pain and discomfort in his hind leg. It appears to be his upper area. When laying to one side his thigh appears to hang funny. The inner of that thigh feels harder than other an feels swollen. Standing up is very hard even rolling into that side. Once up he can extend and will waalk around but limited weight. He has been having an abnormal large amount of hair loss, lowered activity level, and just not the regular happy pup. He is a grumpy old man, getting him into a vehicle to visit a vet will be a very serious challenge in itself so just curious if there’s something to try from home first could it be Cushing
sounds like maybe cushing.There is a cheap test creatinine cortisol ratio urine test might be a good start
No. It is an adrenal disease. Very Different than cancer.
Cushings cannot be cured, but can be treated
Is Cushing disease a form of cancer